We often hear the term “proteins”, and usually use them when talking about food. However, do you know what proteins are? What they are made up of, or what do they do? It is pretty tricky to explain exactly what they are, so we must start from the basics. That said, let us discuss what exactly proteins are.
What are Proteins?
Proteins are biomolecules, so they are essential in making up a part of living things. Proteins always contain carbon, hydrogen, oxygen and nitrogen. They may also contain sulfur.
The monomer of proteins is amino acids, and twenty different amino acids can form peptide bonds between one another to form polypeptides. The sequence of amino acids that make up proteins is determined through the transcription and translation of genes.
Amino Acids
Now, let us look into each amino acid more deeply. Every amino acid contains four Now, let us look into each amino acid more deeply. Every amino acid contains four components bonded to a central carbon atom, including a hydrogen atom, an amino group, a carboxylic group, and a side chain. The side chain is sometimes referred to as an R group, and R groups are variable and are responsible for determining the type of each amino acid.
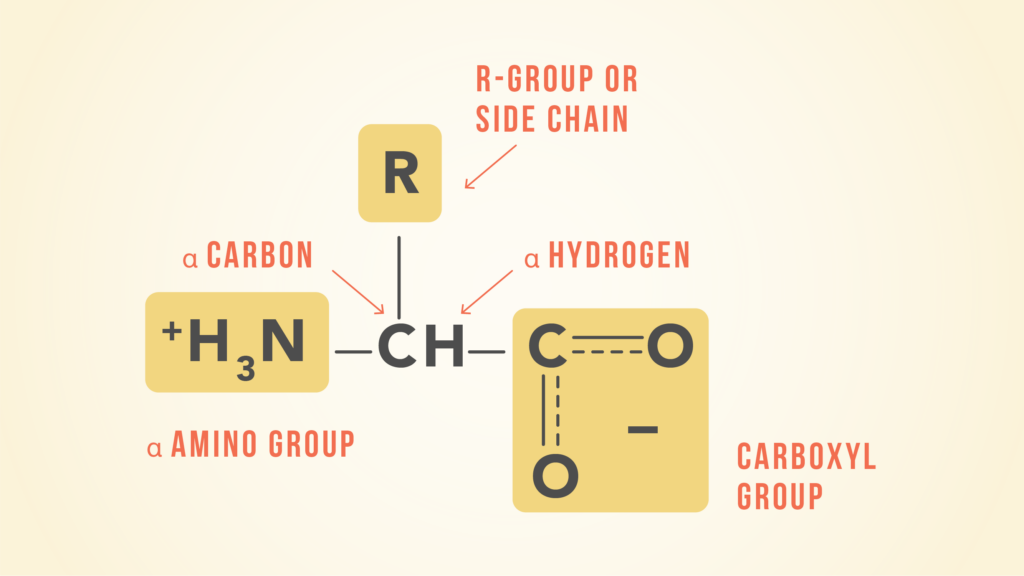
Since R groups determine the type of each amino acid, it also determines their properties. Amino acids can be categorised into four different groups based on their R group – neutral and non-polar, neutral and polar, acidic and polar, and basic and polar.
At this point, you may be wondering what the differences between non-polar and polar R groups are. Polar R groups can form hydrogen bonds with water, making the amino acid hydrophilic. Conversely, non-polar R groups cannot form hydrogen bonds with water, making the amino acid hydrophobic.
Physically, amino acids are colourless, crystalline solids, and they are often soluble in water but not in organic solvents. As amino acids contain a basic amino group and an acidic carboxylic group, they are usually amphoteric, making amino acids suitable buffers when dissolved in a solution.
We can join two amino acids together via a peptide bond in a condensation reaction. This is like the formation of disaccharides in carbohydrates and is similarly known as a dipeptide. Each dipeptide has a free amino end (N-terminus) and a free carboxyl end (C-terminus). In dipeptides and polypeptides, the amino acid with the N-terminus is usually considered the chain’s first amino acid.
Organisation of Proteins
There are four different levels that proteins can be organised into – primary, secondary, tertiary and quaternary. The organisation of proteins is cumulative, which means that tertiary proteins have both primary and secondary structures. It also means that quaternary proteins have all three simpler structures. Most functional proteins have a tertiary or quaternary structure.
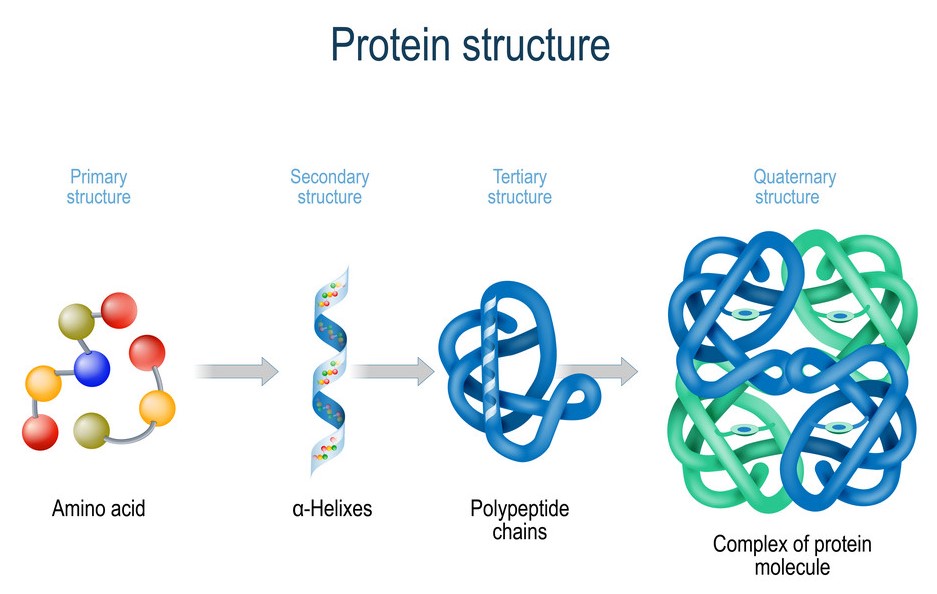
Primary structure
Primary structure refers to the type, number and sequence of amino acids linked by peptide bonds. Every polypeptide has a specific primary structure as it determines its properties, 3D shape, and biological functions.
Secondary structure
Secondary structure refers to localised folds and coils of the polypeptide chain. These folds and coils are due to hydrogen bonds formed between polypeptide chains. The hydrogen bonds are formed at regular intervals along the polypeptide backbone between peptides close to each other. Hydrogen bonds are formed between the oxygen of the carbonyl group of one amino acid and the nitrogen of the amino group of another.
There are two types of secondary structures – α-helices and β-pleated sheets. Both tend to be located within the core of proteins, and hydrophobic R groups of secondary structures are thus shielded from aqueous environments.
The α-helices contain hydrogen bonds formed between the oxygen of one amino acid and the nitrogen of another that is four peptides away. The backbones of α-helices are wound around the long axes, which makes all the hydrogen bonds parallel to the axis and the R groups of every amino acid project outwards. The intra-chain hydrogen bonds help to stabilise the α-helix.
The β-pleated sheets are made up of two or more beta strands of a polypeptide chain. These beta strands are adjacent to each other and are similarly held by intra-chain hydrogen bonds. β-pleated sheets tend to favour hydrophobic R groups due to their amino acid composition. There are parallel and anti-parallel β-pleated sheets.
Tertiary structure
When polypeptides fold extensively upon themselves, forming a precise globular shape, we refer to it as having a tertiary structure. Tertiary structures involve interactions between R groups of amino acids far away from each other. These interactions include hydrogen bonds, ionic bonds, hydrophobic interactions, and disulfide bonds.
As tertiary structure involves folding the polypeptide chain, important peptides that are far away are brought together in localised regions. Localised regions include the active sites, binding sites, and functional sites of the protein. This gives the polypeptide chain a specific 3D conformation.
With the hydrophobic R groups tucked in within the core of the protein, tertiary structures are thus often soluble in water. Hydrophilic R groups are then exposed to aqueous environments.
Quaternary structure
Quaternary structures are formed when two or more polypeptide chains with tertiary structures form bonds with each other. This results in a precise structure between polypeptide units. Proteins such as haemoglobin and collagen have a quaternary structure.
Denaturing Proteins
You might be thinking – now that there are so many different types of bonds that form a protein, how do we break it? Proteins are denatured when their specific 3D conformation is changed. This is caused by unfolding the chain and results in a loss of biological activity. Denaturation affects the secondary, tertiary, and quaternary structure of polypeptides, and it does not affect the primary structures of proteins.
Proteins are denatured under certain conditions.
Changes in temperature
When the temperature increases, amino acid molecules gain kinetic energy, making them vibrate more vigorously. As such, the hydrogen bonds, ionic bonds and hydrophobic interactions are broken. This causes proteins to be denatured.
Changes in pH
Ionic bonds are broken outside of the optimal pH range of the protein. In acidic mediums, H+ ions form bonds with COO– groups of amino acids to form carboxyl groups. In basic mediums, NH3+ groups lose H+ ions to form amino groups. As such, ionic bonds between amino acids of a polypeptide chain are broken.
Others
Proteins can be denatured if there are heavy metal salts in the medium, and this is due to the salts being charged. Reducing agents may disrupt disulfide bonds, resulting in the denaturation of proteins.
Key Proteins
Haemoglobin
Most of us know that haemoglobin is the red pigment found in our red blood cells. More specifically, haemoglobin is an oxygen-carrying globular protein with a quaternary structure. Haemoglobin is compact and comprises four subunits with tertiary structures, and two of these subunits are known as α-chains and β-chains.
Each haemoglobin subunit consists of a haem group, which facilitates the binding of oxygen. As such, haemoglobin is efficient in transporting oxygen around our bodies. The presence of porphyrin rings and iron ions in haem groups allows oxygen to be bound reversibly.
Collagen
We often say that eating food containing collagen would help make our skin smoother. Just how true is this? We first have to find out what collagen is.
Collagen is found in the connective tissues of animals. Our cartilages, ligaments, tendons, bones and teeth contain collagen. It is responsible for skin strength and elasticity and is vital for creating our bodies’ physical structure. Collagen has a quaternary structure and is fibrous.
Collagen is made from many tropocollagen subunits. Each tropocollagen subunit is made up of three helices twisted around each other. Each polypeptide chain has a secondary structure, so collagen exhibits a quaternary structure collectively.
That said, collagen would still be broken down into amino acids when digested, so eating food containing collagen may not make our skin smoother. However, the collagen made by our bodies would help make our skin firmer and more elastic.
Conclusion
Now that we have understood the science behind proteins, we can better understand their function in our daily lives. Of course, many different types of proteins serve many different functions. However, with what you now know about amino acids and the organisation of proteins, you can apply it when learning about new types of proteins.




Add comment