In today’s world, many people are constantly trying to lose weight. Now and then, I often hear something like, “I’m so fat, I need to lose weight” or “I need to cut down on these fats”. Well, losing weight is one thing, but do you know that your body needs these fats as well? These fats are known as lipids. That said, let us discuss what lipids are and what exactly do they do.
What are Lipids?
Lipids, just like carbohydrates, proteins, and nucleic acids, are biomolecules. This means that they play an essential role in the composition of living things. Similar to carbohydrates, lipids are comprised of carbon, hydrogen, and oxygen. However, they do not have a general formula.
There are two types of lipids, fats, and oils, classified into three categories – simple lipids, compound lipids, and steroids and sterols. Based on what we know from our diet, fats are in the solid-state, and oils are in the liquid form. Lipids cannot dissolve in water but are soluble in organic solvents like ethanol. This allows it to be extracted using organic solvents. We will discuss the ethanol emulsion test later.
Structure of Lipids
Lipids are often comprised of glycerol and fatty acids. This is more specific to triglyceride, which we will discuss later.
Glycerol
Glycerol is an alcohol with a molecular formula of C3H5(OH)3. This means that it comprises three hydroxyl groups, which makes it soluble in water. Physically, it is colourless and odourless, tastes sweet, and is not toxic.
Fatty Acids
Fatty acids are aliphatic carboxylic acids, which means that their long hydrocarbon chain can be either saturated or unsaturated. Naturally, these hydrocarbon chains contain an even number of carbon atoms. The hydrocarbon chain of fatty acids is hydrophobic, while the carboxylic acid group at the end of the chain is hydrophilic. The melting point of fatty acids depends on whether they are saturated or not and the length of their hydrocarbon chains.
Saturated fatty acids do not contain C=C double bonds between carbon atoms. The number of hydrogen atoms attached to each carbon atom is maximised. This makes the hydrocarbon chain of saturated fatty acids a straight chain. On the other hand, unsaturated fatty acids contain C=C double bonds between carbon atoms. This creates a kink in the hydrocarbon chain, causing intermolecular forces of attraction. Due to that, unsaturated fatty acid molecules cannot pack closely to one another. As such, saturated fatty acids have higher melting points than unsaturated fatty acids.
For unsaturated fatty acids, the presence of C=C double bonds gives rise to cis-trans isomerism. While most unsaturated fatty acids occur naturally as cis isomers, there are many examples of fatty acids with trans isomerism. One notable example is the trans isomer of oleic acid, elaidic acid.
Types of Lipids
As mentioned earlier, there are three general types of lipids – simple lipids, compound lipids, and steroids and sterols. Simple lipids are formed when fatty acids are bonded to an alcohol group via ester bonds. An example of an alcohol would be glycerol, where three fatty acid molecules are bonded. Complex lipids are made up of lipid and non-lipid components. These include phospholipids and glycolipids.
Now, for steroids and sterols. Sterols are steroids with a hydroxyl group in their nucleus. All of them have a carbon skeleton with four fused rings. Examples of steroids and sterols include cholesterol, testosterone, and oestrogen.
Triglycerides
There are many different types of triglycerides out there. These include triglycerides with saturated or unsaturated fatty acids. They are found in many different parts of living things, including our body fats, blood, and vegetable oil. Despite being insoluble in water, the components of triglycerides can mostly dissolve in water.
Structure
Triglycerides are esters formed by attaching three fatty acid molecules to a glycerol molecule via ester bonds. When fatty acid molecules form ester bonds, a water molecule is removed, making it a condensation reaction.
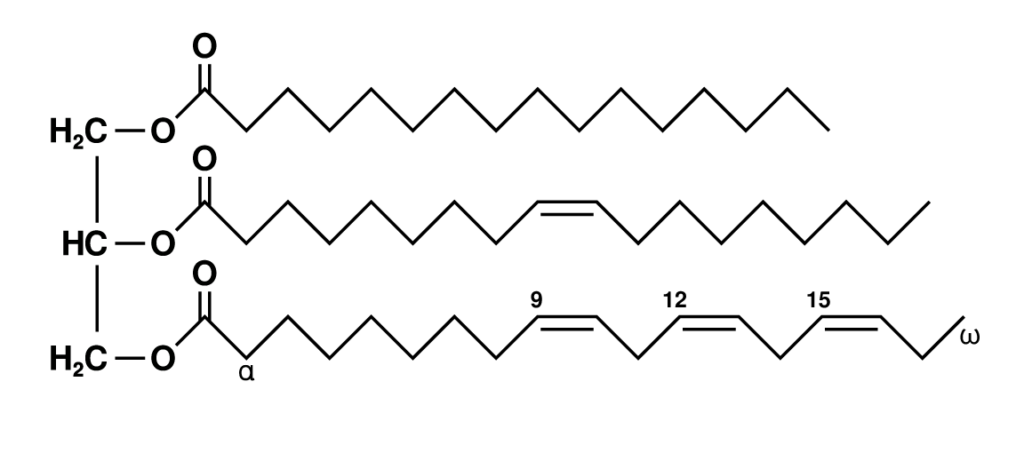
Functions
The critical functions of triglycerides are storing metabolic energy and metabolic water. As triglycerides are large insoluble molecules, many can be stored without affecting the water potential. As a good storage form of energy, triglycerides give out more energy than glycogen alone or starch. On top of that, they will not diffuse out of the cell. Lipids are suitable for animals like migratory birds or hibernating animals.
Triglycerides can store energy well, but how does it keep the water well? When hydrogen atoms of the triglyceride molecules are oxidised, they form water. Since triglyceride molecules contain more hydrogen atoms than carbohydrates, they can store more water.
Triglycerides are also good thermal insulators and can help increase the buoyancies of aquatic animals. This is due to its density being less than that of water. On top of that, triglycerides can also serve as a protective layer to protect critical internal organs, such as our kidneys.
Phospholipids
Phospholipids are complex lipids that are amphipathic. This is due to the hydrophobic properties of hydrocarbon chains and the hydrophilic properties of the phosphate group. On top of that, small molecules that are often polar or charged can be attached to the phosphate group of phospholipids. Phospholipids are found in cell membranes.
Structure
Like triglycerides, fatty acid molecules bond to the glycerol molecule via ester bonds to form phospholipids. A negatively charged phosphate group is attached to the remaining hydroxyl group of the glycerol via a phosphoester bond. However, phospholipids only have two instead of having three fatty acid molecules.
Functions
Just like what we have mentioned, phospholipids are found in cell membranes. This includes the cell surface membranes and the membranes of different organelles within the cell. Phospholipids make up the basic bilayer of membranes.
Cholesterol
Cholesterol is categorised under sterols. They play an essential role in animal cells, including the aforementioned cell membrane. We often hear about people having “high cholesterol levels” or “low cholesterol levels”. We are talking about this same cholesterol.
Structure
As a sterol, cholesterol has a carbon skeleton with four fused rings. It also has a molecular formula of C27H46O.
Functions
Cholesterol is crucial for life in animals, which explains why having too much or too little cholesterol poses issues to your health. They provide mechanical stability to our cell membranes and maintain the fluidity of the membranes. On top of that, cholesterol minimises uncontrolled leakage from cells.
Ethanol Emulsion Test
The ethanol emulsion test determines whether lipids are present in a sample. Ethanol is added to the sample in a test tube, then shaken thoroughly. If the solution remains colourless, lipids are not present in the sample. However, if you see a layer of cloudy white suspension, there are lipids in the sample.
Conclusion
We have mentioned a lot about lipids! However, one thing is for sure – lipids are not necessarily harmful to your health. However, too much of it is not beneficial to your health. Understanding lipids will help you understand how to manage your diet better to control your lipid intake.





Add comment